Our primary focus involves the construction of sophisticated in vitro models representing human pathology. Employing cutting-edge micro- and nanotechnology, we manipulate biological components at a scale relevant to both physiology and pathology. The ultimate objective is to uncover the fundamental mechanisms underlying diseases, enhance disease diagnosis, and innovate in the development of novel therapies.
Sickle Cell Disease
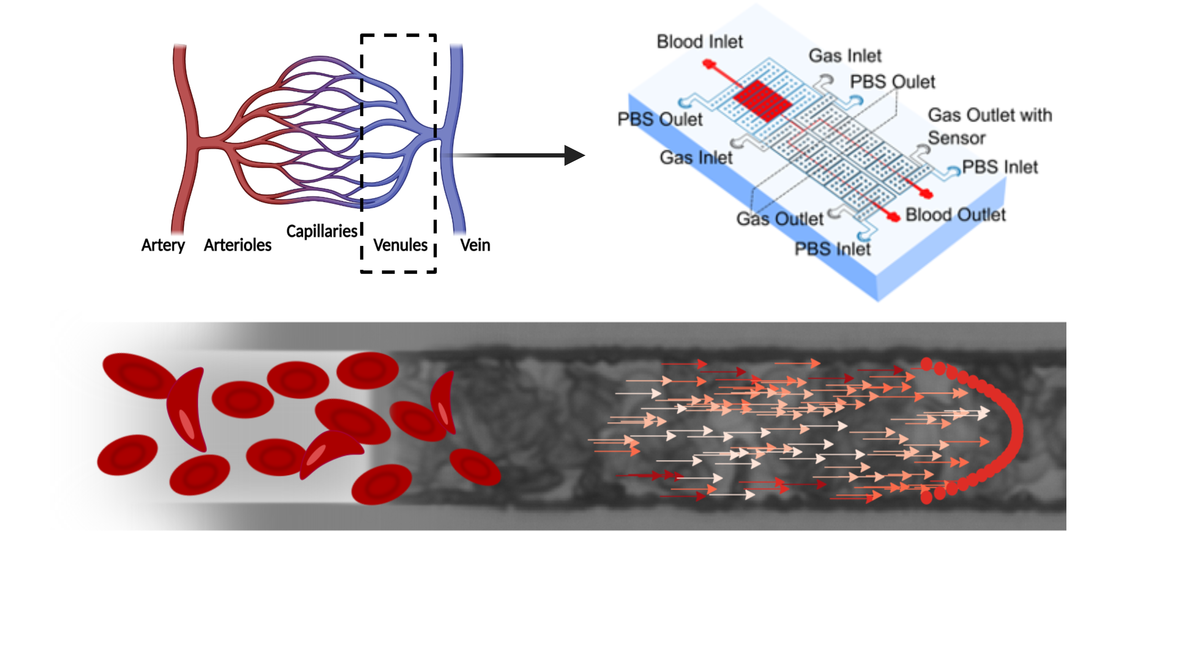
Sickle cell disease is a devastating genetic disorder that affects more than 20 million people worldwide and results in chronic pain, fatigue, frequent hospitalization, and high mortality, especially among children. A major focus of our group is to understand the mechanisms of vaso-occlusion, the primary pathological mechanism in sickle cell disease.
We have developed a range of tools and methods to facilitate these studies, including physiologically sized microfluidic channels, tools to regulate blood pressure, methods to finely control blood oxygen, and methods to monitor blood flow in real time. Our goals are to understand how vaso-occlusion proceeds in vivo and under which conditions vaso-occlusions are most likely to occur. Furthermore, we aim to predict high-risk patients for vaso-occlusive crises and leverage the knowledge and tools developed in this pursuit to drive the discovery and development of new therapies for this debilitating disease.
Cancer Metastasis
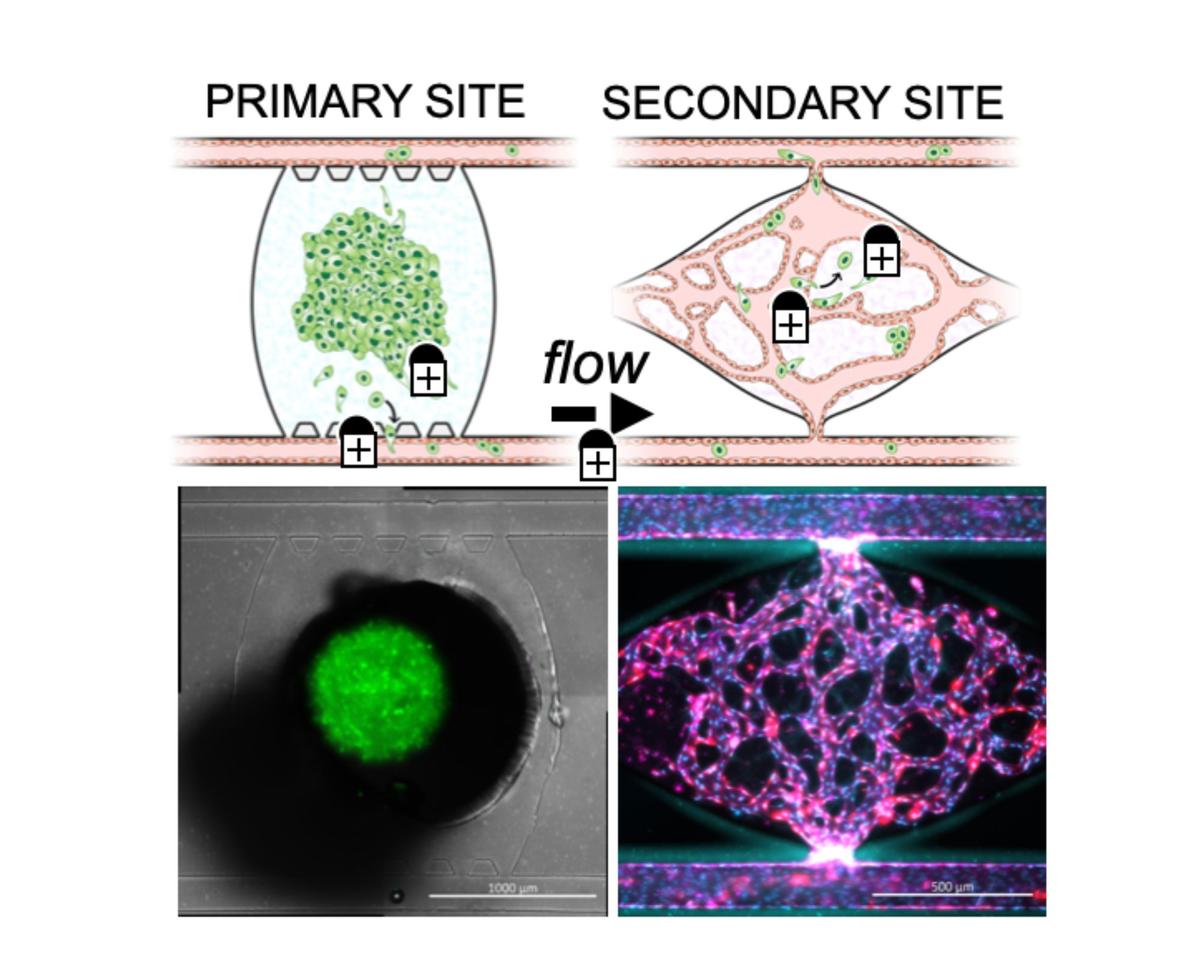
Metastases account for approximately 90% of human cancer-related deaths, yet our understanding of metastasis stages and mechanisms remains limited. Our laboratory is developing a novel microfluidic platform to directly visualize and study the metastatic process, including late stages like extravasation and colonization, as well as dormancy and the premetastatic niche. This platform enables dynamic temporal analysis in a physiologically relevant context, offering insights into tumor cell features and the metastatic niche.
Our goal is to address key questions regarding cell dormancy, the metastatic environment, the tumor microenvironment, and the premetastatic niche, along with the cellular interactions in these environments. We aim to deepen our understanding of the complexities of metastatic and dormant environments and aid in the development of potential metastasis-blocking therapies.
3D Tissue Culture
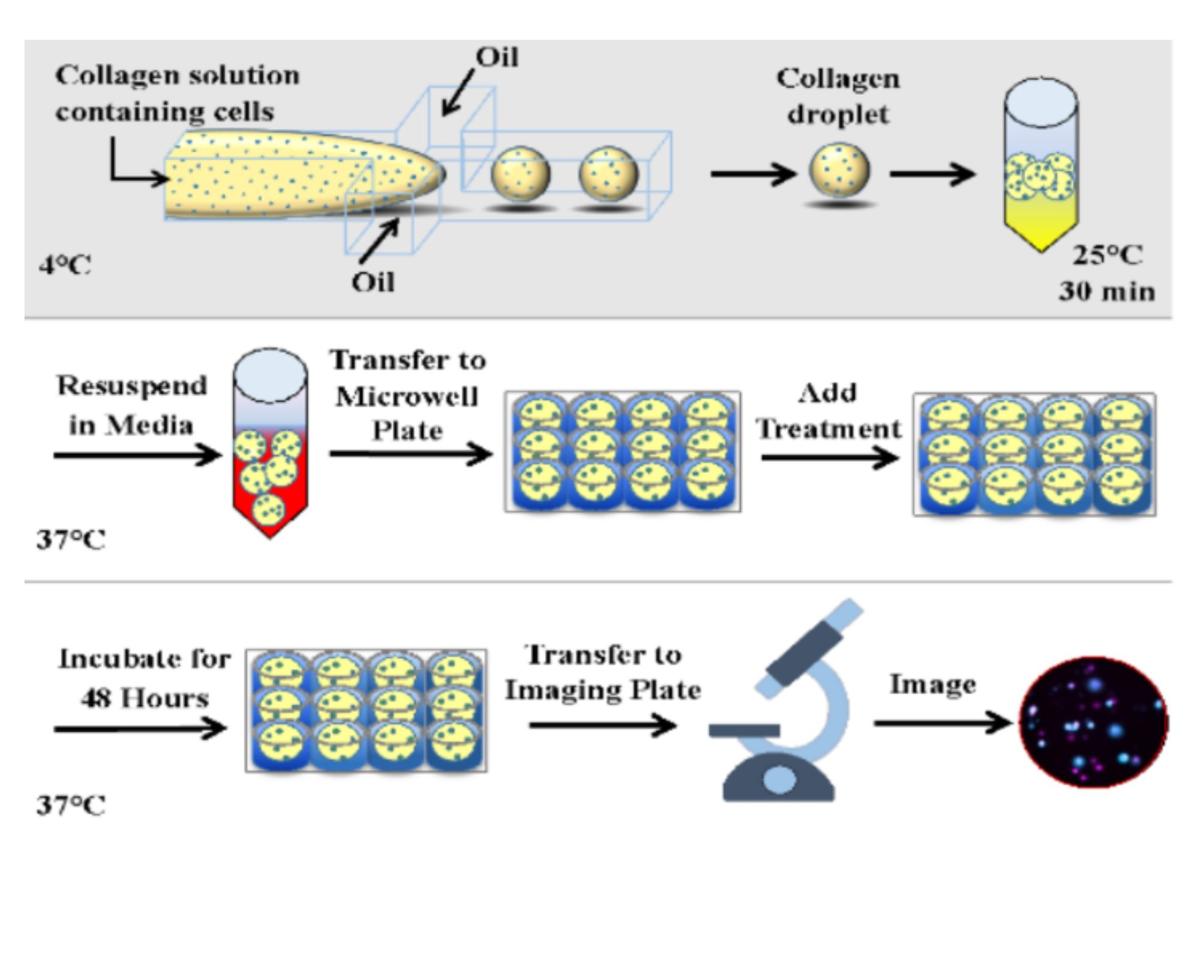
Drug discovery heavily relies on in vitro models for initial drug testing, aiming to predict clinical trial outcomes. However, these models often fail to accurately represent human organs and diseases, leading to high rates of late-stage trial failures, increasing time and costs. To address this, our group is developing a high-throughput 3D tissue culture platform using microfluidic droplet technology. This platform generates microscale tissue constructs with high biological complexity, enabling rapid assays of cellular functions and drug responses.
Our aim is to develop more physiologically relevant models by harnessing cutting-edge micro- and nanotechnology. This allows us to manipulate biological components at a scale that is relevant to both normal physiology and pathological conditions. By leveraging this technology, we seek to enhance drug testing and biological research, ultimately advancing our understanding of diseases and the development of therapies.